This web page was produced as an assignment for Genetics 677, an undergraduate course at UW‐Madison.
Protein Structure and Localization
Protein 3D structure
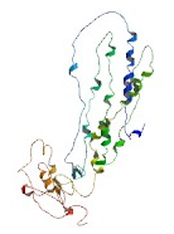
Partial 3D structure of human ABCA1 [1]
One of the best methods for determining a protein's 3D structure is X-ray crystallography, but until crystallography is performed, predictions of protein secondary and tertiary structures can be made based on conserved primary structures (DNA/amino acid sequences).
The image to the right is a detailed model of an ABCA1 domain from the human ABCA1 protein [1]. The model is the result of many independent studies to determine amino acid sequence, post-translational modifications and protein interactions.
Many domain-prediction tools, including the ones used below, are free to the public.
The image to the right is a detailed model of an ABCA1 domain from the human ABCA1 protein [1]. The model is the result of many independent studies to determine amino acid sequence, post-translational modifications and protein interactions.
Many domain-prediction tools, including the ones used below, are free to the public.
Structure and Localization Links
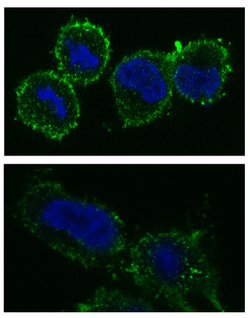
Where does ABCA1 act in the cell?
The ABCA1 protein is present in the cell plasma membrane of most cells, but it is most highly expressed in the liver and intestinal epithelial
cells [2].
cells [2].
Abnormal localization
in Tangier disease
Tangier disease-related ABCA1 mutant proteins show deficient localization to the plasma membrane [4]. This suggests that Tangier disease is ultimately caused by mutations that prevent ABCA1 from reaching the membrane [4].
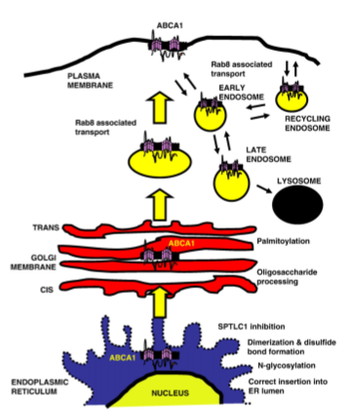
The Localization Process
At the endoplasmic reticulum (ER), ABCA1 undergoes dimerization before being exported to the golgi body for processing. In a poorly understood process, the protein must then reach the plasma membrane [5].
Once ABCA1 reaches the plasma membrane, it forms a homotetramer and can then perform its transporter activities [5].
Once ABCA1 reaches the plasma membrane, it forms a homotetramer and can then perform its transporter activities [5].
Sources:
1. http://modbase.compbio.ucsf.edu/modbase-cgi/index.cgi
2. http://www.uniprot.org/uniprot/O95477
3. http://www.antibodies-online.com/antibody/337112/anti-ATP-Binding+Cassette+Sub-Family+A+Abc1+Member+1+ABCA1+aa1800-2260/#image
4. Cameron J, Ranheim T, Halvorsen B, Kulseth M, Leren T, Berge K. Tangier disease caused by compound heterozygosity for ABCA1 mutaions R282X and Y1532C. Atherosclerosis. 2010; 209: 163-166.
5. Kang MH, Singaraja R and Hayden MR. Adenosine-Triphosphate-Binding Cassette Transporter-1 Trafficking and Function. Trends in Cardiovascular Medicine. 2010; 20(2); 41-49.
1. http://modbase.compbio.ucsf.edu/modbase-cgi/index.cgi
2. http://www.uniprot.org/uniprot/O95477
3. http://www.antibodies-online.com/antibody/337112/anti-ATP-Binding+Cassette+Sub-Family+A+Abc1+Member+1+ABCA1+aa1800-2260/#image
4. Cameron J, Ranheim T, Halvorsen B, Kulseth M, Leren T, Berge K. Tangier disease caused by compound heterozygosity for ABCA1 mutaions R282X and Y1532C. Atherosclerosis. 2010; 209: 163-166.
5. Kang MH, Singaraja R and Hayden MR. Adenosine-Triphosphate-Binding Cassette Transporter-1 Trafficking and Function. Trends in Cardiovascular Medicine. 2010; 20(2); 41-49.